Gene Editing and Regulation
Gene Editing
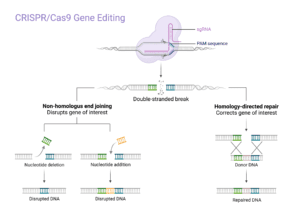
Gene editing is a technique that allows scientists to alter the DNA of living organisms. The most common tool to introduce these alterations is the clustered regularly interspaced short palindromic repeats (CRISPR). CRISPR was originally extracted from bacteria, where it helps protect from foreign invaders, such as viruses. This tool was later used to alter genes in mammalian cells with significant promise.
Gene Regulation
Another way to regulate genes is using RNA interference. This technique uses small RNA segments to interfere with the expression of specific genes. This can be used to turn off genes or reduce the amount of proteins expressed by them. RNA interference has been used to study the function of different genes, and to treat diseases like cancer and hepatitis C.
The discovery of CRISPR
CRISPR is part of the adaptive immune system in the archaea and bacteria where it is responsible for defending against foreign nucleic acids, introduced via plasmids and phages (1). Even though the acronym CRISPR was introduced in 2002 (2, 3), the immunologic rules of these repeated DNA sequences were discovered in 2005 by Mojica and colleagues (4). Following that, Charpentier and Doudna reported the mechanism and biomedical characteristics of the CRISPR-associated protein 9 (Cas9) (5). However, the most significant breakthrough was in 2015 when Zhang was successful in adopting the CRISPR-Cas9 in eukaryotic cells for targeted genome editing (6). The major benchmarks of the development of CRISPR systems are summarised here (7).
The inner workings
The potential of CRISPR
The rise of CRISPR has revolutionised gene editing, which holds vast potential for treating genetic diseases. CRISPR is a simple, efficient, and precise tool for modifying genes. It has already been used to treat a number of diseases in humans and animals. The ability to precisely edit genes has the potential to cure many genetic diseases, including cystic fibrosis, sickle cell anemia, and muscular dystrophy. CRISPR may also be used to treat cancer by disabling the genes that allow tumours to grow, among other diseases that originate from genetic mutations.
Introduction
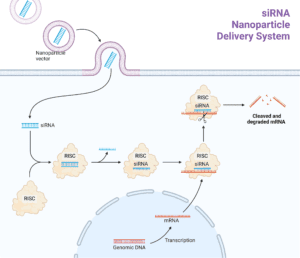
With the development of gene-editing technologies, researchers have begun exploring how to harness the potential of RNA interference (RNAi) for therapeutic purposes. RNAi is a natural process in which double-stranded RNA molecules (also known as siRNAs) are used to silence genes within cells by binding with them and preventing the translation of the messages they carry. When these siRNAs are delivered into a cell in their unaltered form, however, they’re often quickly broken down by enzymes that recognize the fact that they contain nucleotides arranged into three-dimensional structures not normally seen inside the cells.
What is gene regulation using siRNA
In order to understand gene regulation using siRNA, it is important to first understand how genes are regulated.
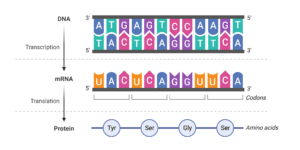
Genes in the human body are transcribed into RNA by a process called transcription. This can be seen as a template for proteins that will be created during translation. The protein is made up of amino acids linked together. The sequence of these amino acids determines what each protein will do and therefore how it will affect human health (or disease!). siRNA can block the expression of an mRNA molecule by preventing its translation into protein or by facilitating its degradation by cellular processes known as RNA interference or RNAi (for short) (figure 1).
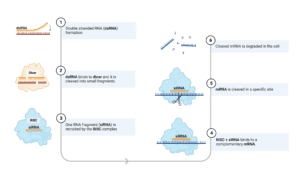
Short interfering RNA can block the expression of a target gene.
siRNA can be designed to bind to messenger RNA (mRNA) and prevent it from being translated into protein. This process is called gene silencing because siRNA prevents the expression of a target gene. To be effective in the body, siRNA has to be protected from enzymes that break it down. To achieve this, siRNA must be either modified or coated with specialised delivery platforms that prevent it from being broken down.
The use of lipid nanoparticles to deliver siRNA
While RNAi is a promising technology for the treatment of many health conditions, delivery of siRNA has been a major challenge. However, efforts to overcome this obstacle have shown promising results and are currently in clinical trials. One example is Onpattro (also known as Patisiran), which was approved by the US Food and Drug Administration (FDA) in August 2018 for the treatment of polyneuropathy caused by an illness called hereditary transthyretin-mediated amyloidosis (hATTR amyloidosis). This drug consists of lipid nanoparticles coated with polyethylene glycol (PEG). Read more about Lipid Nanoparticles (LNPs) here.
Introduction
mRNA is a molecule that contains both genetic information and its translation process. The mRNA molecule can be used to create proteins, which are responsible for many biological processes (figure 1). This is a versatile method of gene therapy that can be used in several different approaches. It can be used to improve the immune system or control disease symptoms; it could also help treat cancer or even aid in the development of vaccines.
The history of mRNA therapeutics.
It should be noted that the use of mRNA therapeutics is not new. The first therapeutic use of mRNA was made in the 1960s, when scientists isolated it from cells and injected it into animal models to fight cancer. This early work led to the development of a number of drugs based on similar principles over the next several decades. However, it is only in the last couple of years, that these products have finally progressed beyond the pre-clinical phase to reach the clinic. Their clinical development has been met with a variety of challenges including manufacturing issues and reproducibility concerns around the delivery methods. Nonetheless, there is much optimism regarding these products as they have shown great promise in pre-clinical studies and have begun to enter clinical trials.
The use of mRNA therapeutics has been expanded into vaccines and immunotherapeutic applications. The potential applications for mRNA therapeutics include treatment for multiple diseases such as cancer, inflammation, liver disease and cardiovascular diseases. More recently, the use of mRNA achieved great success in the development of vaccines against COVID19.
mRNA in vaccine development.
The introduction of mRNA molecules has successfully been used to generate viral antigens directly within host cells. One of the first clinical trials using this approach has shown that intramuscular injection of mRNA encoding H5N1 hemagglutinin (HA) can elicit an immune response in a patient without any adjuvant being required. This strategy results in high-level expression of HA, which is then converted into immunogenic peptides that can be recognized by antibodies and induce neutralizing antibody activity against H5N1 viruses. Similarly, studies have shown that administering a single dose of mRNA encoding SARS-CoV surface glycoprotein (S) raised neutralizing antibody titers against both SARS-CoV and severe acute respiratory syndrome coronavirus (SARS-CoV). The development of mRNA-based vaccines has enabled the use of immunogens to be rapidly adapted to new viral targets and has successfully produced immunogenic vaccines against viruses such as SARS-CoV-2, influenza and kindles with a rapid response time.
Drug delivery of mRNA using Lipid nanoparticles
The use of mRNA in therapeutics and vaccines presents a unique challenge: the instability of mRNA when it is delivered on its own. To overcome this challenge, researchers have developed a method called lipid nanoparticle delivery (LNP), which uses these particles as carriers for mRNA (figure 2). In particular, LNPs have been shown to be successful in delivering COVID19 vaccines like Pfizer and Moderna’s next-generation vaccine against SARS-CoV-2 (COVID19). In addition to demonstrating higher efficacy than other delivery methods such as intradermal injection or intramuscular injection (which are often used for other non-invasive methods).
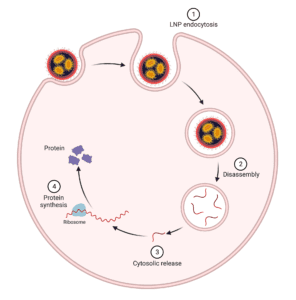
Conclusion
We believe that the use of mRNA as a vaccine or therapeutic vector will continue to grow, as this approach has several advantages over traditional protein-based vaccines. It is also likely that there will be many more applications of mRNA in the future, with new targets being explored by both academia and industry. Prorenata Biotech offers a range of services for the design and development of your target mRNA therapeutics and vaccines, including identification, designing, testing, and developing specialised delivery platforms.
- Ishino Y, Shinagawa H, Makino K, Amemura M, Nakatura A. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isoenzyme conversion in Escherichia coli, and identification of the gene product. Journal of Bacteriology. 1987;169(12):5429-33.
- Jansen R, Van Embden JDA, Gaastra W, Schouls LM. Identification of genes that are associated with DNA repeats in prokaryotes. Molecular Microbiology. 2002;43(6):1565-75.
- Jansen R, van Embden JD, Gaastra W, Schouls LM. Identification of a novel family of sequence repeats among prokaryotes. Omics : a journal of integrative biology. 2002;6(1):23-33.
- Mojica FJM, Díez-Villaseñor C, García-Martínez J, Soria E. Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements. Journal of Molecular Evolution. 2005;60(2):174-82.
- Deltcheva E, Chylinski K, Sharma CM, Gonzales K, Chao Y, Pirzada ZA, et al. CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature. 2011;471(7340):602-7.
- Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339(6121):819-23.
- Siva N, Gupta S, Gupta A, Shukla JN, Malik B, Shukla N. Genome-editing approaches and applications: a brief review on CRISPR technology and its role in cancer. 3 Biotech. 2021;11(3):146.
- Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012;337(6096):816-21.
We offer a range of services for the identification of target genes and the development of specialised approaches to edit and regulate them. Whether you are looking for new targets to edit or need help developing an approach to achieve specific results, we are here to help achieve your desired outcome.
The second step in the process development is selecting and designing the right tools and components of your therapeutics. We will determine the right CRISPR variants from our extensive toolbox, select the most efficient gRNA sequences, design novel siRNA constructs using cutting-edge technologies, or map the right pDNA for cell line development.
Testing is an essential part of the development journey. We work with you to either develop a new cell line or test the designed platform on specific cell lines, depending on your needs. We will then select the top candidate components for the final validation.
We use several approaches for validating the success of the designed platform. Depending on the design, we will validate the outcomes with sequencing, profiling mRNA expression, or testing for protein expression. This will help us to confirm that the platform is working as intended and producing the desired results.
Prorenata Biotech is specialised in drug delivery for genetic medicine. We design and develop scalable Lipid Nanoparticle (LNPs) solutions that are tailored to specifically meet your requirements. Our LNPs are designed from the early preclinical stages, all the way to GMP-grade LNPs for clinical applications.